Natchez Hanson considers one of her proudest accomplishments walking across the stage to receive her college diploma – earning a bachelor’s degree in math education. Hansen, 24, now a high school math teacher in Polk County, lives daily with the challenges of Friedreich’s ataxia.
Friedreich’s ataxia, a rare debilitating neuromuscular disease, typically strikes between the ages of 5 and 15, causing vision, balance, speech and cardiac problems and progressively robbing a young person of their energy, strength and ability to walk.
While physical therapy helps Hanson work on core strength and balance, there is no approved treatment for Friedreich’s. That’s why she is so excited to be part of a USF-led national clinical trial of a drug that researchers, clinicians and patents hope will be the first to improve the symptoms of the life-shortening disease.

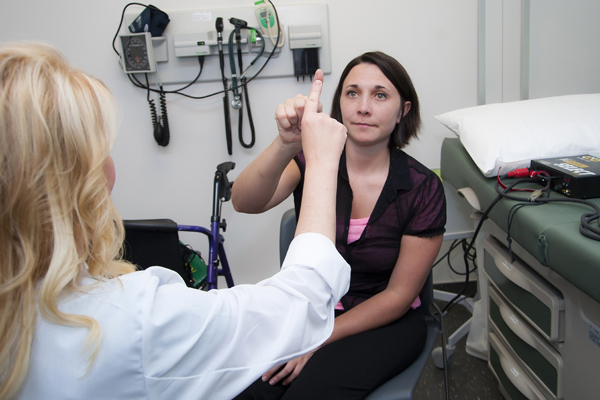
USF Health neurologist Dr. Theresa Zesiewicz, left, with patient Natchez Hanson, one of the participants in the USF-led national clinical trial for a potential Friedreich’s ataxia drug.
“I don’t want anyone else to feel the way I did (when I was diagnosed) if they don’t have to,” Hanson said. “I just cried because it was scary. A lot of people died being really young, and I was only 17. I had all these dreams… I just wanted to be normal.”
The double-blind, placebo-controlled trial, sponsored by Edison Pharmaceuticals, Inc., in collaboration with the Friedreich’s Ataxia Research Alliance (FARA), is led by neurologist Dr. Theresa Zesiewicz, director of the USF Ataxia Research Center.
Researchers are primarily testing the effectiveness of the investigational drug EPI 743, a potent antioxidant, on vision, in patients with Friedreich’s ataxia, many of whom experience varying degrees of visual changes. Secondarily, the study will evaluate neurological function.
Sixty patients with a genetically-confirmed Friedreich’s ataxia diagnosis have been enrolled in the study, which involves Children’s Hospital of Philadelphia at the University of Pennsylvania and UCLA in Los Angeles, CA, as well as lead site USF.
When the study was announced at last year’s USF/FARA Scientific Symposium, the news was welcomed with cheers and tears by patients and their families who had come to learn about the latest advances in ataxia research.
The reaction and subsequent overwhelming international interest by prospective trial participants did not surprise Dr. Zesiewicz and others who care for those with Friedreich’s ataxia.
“These are young people – children, teens, young adults – affected by a relentless disease that can cause early death,” said Dr. Zesiewicz, USF Health professor of neurology. “There is a real sense of urgency to find a first treatment … We’re racing against time here.”
EPI-743 isn’t the only drug in the pipeline for Friedreich’s ataxia and related disorders, but it has shown promise in some early, limited trials — including a study showing neurological benefit in children with Leigh syndrome, a mitochondrial disease, like Friedreich’s, that attacks energy metabolism. And, so far at least, the drug appears to be well tolerated.
EPI-743 is an extremely powerful antioxidant — much stronger than the vitamin E you can get at a drugstore, Dr. Zesiewicz said. “The thinking is that this very powerful antioxidant will work on the energy-producing part of the cell, the mitochondria, to improve symptoms.”
While Friedreich’s ataxia is the focus of the USF-led study, Dr. Zesiewicz said, “EPI-743 may have implications for other neurodegenerative disorders like Parkinson’s disease and Alzheimer’s disease.”
USF is one of 10 partners in the FARA Collaborative Clinical Research Network, an international network of centers sharing and resources to advance treatments and clinical research for people with Friedreich’s ataxia.
The USF Ataxia Research Center conducts several studies in addition to the EPI-743 trial, including a clinical study on cardiac dysfunction in Friedreich’s ataxia and another looking for biomarkers to better monitor disease progression.
Edison Pharmaceuticals CEO Dr. Guy Miller, said USF was the logical choice to take the lead role in the latest EPI-743 trial.
“Many centers have silos of excellence in basic science, or clinical research, or translational research, but at USF there is a cross-section of excellence in all three, which is essential for conducting a well-run 2B clinical trial. ” Dr. Miller said. “You also have a fantastic alignment of physician leaders who are innovative and care deeply about patients.”

L to R: Dr. Stephen Klasko and Dr. Zesiewicz with Dr. Guy Miller, CEO of Edison Pharmaceuticals, at last year’s USF/FARA Scientific Symposium, where Dr. Miller announced the EPI-743 clinical trial for Friedreich’s ataxia. Dr. Miller returned to this year’s symposium, giving an update on drug discovery innovation and progress.
Dr. Clifton Gooch, chair of USF Health Neurology and director of the USF Neuroscience Collaborative, emphasized both the rapid rise of the center and the prospects for EPI-743.
“The USF Ataxia Research Center has become a leading international clinical trials center in just a few short years under Dr. Zesiewicz’s dedicated leadership, and we are excited to now be testing one of the most promising drugs yet tried for the treatment of FA,” he said.
Friedreich’s ataxia is a caused by defects in the gene carrying instructions for a protein called frataxin, which leads to diminished energy production in cells, including those of the nervous system and heart. While rare – only one in 50,000 people are affected by the inherited disease – one in 100 carry the gene, many without even being aware of it.
“Mom and Dad carry the Friedreich’s ataxia gene, but neither one knows that they carry the gene, so it’s often a big surprise to the parents when the child is diagnosed,” Dr. Zesiewicz said.
For many children the first sign that something is physically wrong are falls. That’s what Hanson remembers.
“When I was growing up, I always walked a little goofy,” she said. “I would run and fall down, but then I’d just get right back up and start running again.”
As time progressed she had trouble walking in a straight line, and leaned on friends and family for support, eventually graduating to crutches. After physicians ruled out several other potential causes, including a brain tumor, Hanson was formally diagnosed with Friedreich’s ataxia at age 17. She started using a walker two years ago.
At first, Hanson said, she was embarrassed by the walker, but now she embraces the greater mobility it provides and uses its storage compartment to help carry school supplies. “At least no one thinks I’m drunk anymore, so that’s good,” she quips.

Results of the Edison EPI-743 trial are expected later next year. In the meantime, Hanson continues to move forward, refusing to let a disease stop her. For the third consecutive year, she was among the patients who spoke Sept. 5 at the 5th Annual USF-FARA Scientific Symposium — sharing their stories of living as optimistically as possible with Friedreich’s ataxia.
“You only have one life, and everyone has a burden to bear. You either laugh about it or you cry about it – and I’m done crying,” she said. “I’m trying to be as happy as I can be.”
See related story: USF/FARA scientific symposium to share latest advances in ataxia research

Photos by Aimee Blodgett, USF Communications and Marketing
