A Washington University-USF Health preclinical study finds that the novel nanoparticle delivers gene-level treatment to suppress KRAS-driven cancer without adverse effects
Despite advances in cancer survival, more than 90 percent of people with pancreatic cancer die within five years. Most patients with pancreatic tumors (and half of those with colorectal cancers) carry a mutation in the KRAS gene, which normally controls cell growth and death.
The KRAS oncogene was discovered more than 35 years ago and is considered one of the most desirable targets in cancer biology — particularly for cancers (like pancreatic) often diagnosed late and in desperate need of improved therapies to prolong survival. Yet KRAS has earned a reputation as being “undruggable” by researchers who continue searching for effective ways to inhibit the mutated form of RAS proteins driving the growth of deadly tumors.
Now a preclinical study led by Washington University School of Medicine in St. Louis, including co-investigators at the University of South Florida Health (USF Health) Morsani College of Medicine, Tampa, Fla., has demonstrated that specially designed peptide-based nanoparticles can suppress pancreatic cancer growth without the toxic side effects and therapeutic resistance seen in drug trials. The findings were published July 30 in Oncotarget.
The nontoxic, peptide-based p5RHH nanoparticles designed by USF Health researchers Samuel Wickline, MD, and Hua Pan, PhD, MBA, deliver an RNA molecule known as small interfering RNA, or siRNA (also known as silencing RNA). The molecules silence the chemical signal (message) telling the KRAS oncogene to make more mutated KRAS proteins that cause pancreatic cells to grow uncontrollably and largely resist existing cancer-killing drugs.
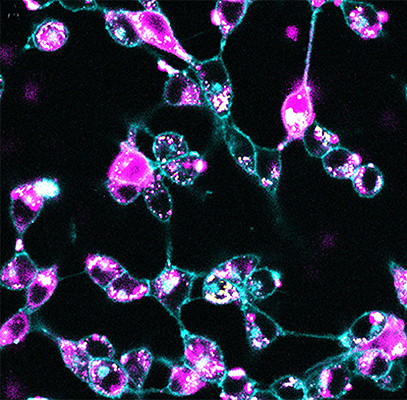
Confocal microscopy image shows fluorescent-tagged nanoparticles (pink) carrying siRNA diffusely taken up by mouse colorectal cancer cells.| Photo courtesy of Washington University School of Medicine in St. Louis
“We’ve developed a nanoparticle system that gets enough of the therapeutic molecule from the bloodstream to the tumor cell without (the molecule) being metabolized or excreted,” said coauthor Dr. Wickline, professor of cardiovascular sciences and director of the USF Health Heart Institute. “The nanoparticle is actively taken up by the targeted tumor cells and then the molecule escapes and does its job to prevent production of mutated KRAS proteins.”
“Approaches that precisely target tumors with various therapies are the future of cancer care. Nanoparticle delivery allows higher concentrations of drugs to reach their target while sparing normal tissues any side effects,” said senior author Ryan Fields, MD, professor of surgery and chief of surgical oncology at Washington University School of Medicine. “In pancreatic cancer, where breaking through the resistant tumor microenvironment is a current unmet need, this approach has the potential to improve therapeutics and patient outcomes.”
Compared to control cells, nanoparticle treatment of both pancreatic and colorectal cells was shown to deliver KRAS-specific siRNA, decrease KRAS RNA expression and lead to increased tumor cell death. Using a genetically engineered mouse model for spontaneously arising pancreatic cancer, the researchers also demonstrated that the intravenously administered nanoparticles could selectively target silencing RNA against the KRAS oncogene to ultimately slow KRAS-driven pancreatic cancer growth. Their system effectively delivered this nanotherapy even in the “stroma-rich” environment of pancreatic tumors.

3D reconstruction of a single pancreatic cancer cell treated with siRNA nanoparticles. Confocal microscopy depicts accumulation of strong fluorescent silencing RNA signaling (pink) within the cell membrane (cyan) boundaries, but separate from lysosomes (yellow) that can degrade the nano-delivered molecules. | Photo courtesy of Washington University School of Medicine in St. Louis
Pancreatic cancer is so difficult to treat in part because fibrous tissue, or stroma, that surrounds the solid tumor “like the shell of a clam” is much denser than the stroma surrounding other more treatable tumors, Dr. Wickline explained. This protective stromal barrier can complicate nanoparticle-delivered treatment.
“Our p5RHH peptide nanoparticle is relatively small and it can squeeze in and out of tight spaces to get (treatment) safely into tumor-specific cells while staying out of normal tissue,” he said. “It avoids adverse ‘off-target’ effects.”
Cancer is very efficient at evading treatments that target one, or even a few mutated genes or proteins, Dr. Wickline said. “The advantage of this nanoplatform carrying siRNA is that it’s easy to change out the target you want silenced, or add many more targets for simultaneous treatment in the same tumor cell.”
The nanoparticles formulated by Dr. Wickline and Dr. Pan have also shown promise for siRNA treatment in mouse models of atherosclerosis and arthritis.
The Oncotarget reported study was supported in part by grants from the National Cancer Institute.


