The USF Health neurobiologist focuses on understanding genetic risk factors that may offer new therapy targets to delay or protect against age-related cognitive decline
There has been a steep rise in the number of Americans dying of Alzheimer’s disease – up 145 percent between 2000 and 2017 The burden of this neurodegenerative disease, which relentlessly diminishes the mind, is not only borne by those living more years in a state of disability and dependence before dying, but by the family members who care for them.
No treatments exist to cure or slow the progression of Alzheimer’ disease, the major form of dementia afflicting an estimated 5.8 million Americans.
“The goal of our research is to reduce the (brain) pathology leading to Alzheimer’s disease, by identifying targeted treatments to delay the onset of disease and protect cognitive function,” said Gopal Thinakaran, PhD, professor of molecular medicine and associate dean for neuroscience research at the USF Health Morsani College of Medicine. “Finding ways to extend cognitive function so that an older person is still able to continue their daily activities or recognize a loved one – even for five more years – would greatly benefit both those suffering from Alzheimer’s and their families or other caregivers.”

Gopal Thinakaran, PhD (center), who holds the Bagnor Endowed Chair in Alzheimer’s Research, with his research team at the USF Health Byrd Alzheimer’s Center.
Dr. Thinakaran, an internationally recognized Alzheimer’s disease researcher, joined the University of South Florida from the University of Chicago in August to help accelerate the interdisciplinary work of the USF Health Neuroscience Institute. That includes recruiting a critical mass of basic scientists who can complement the university’s ongoing Alzheimer’s research while also expanding efforts to translate laboratory findings into new therapies for other neurodegenerative disorders, including Parkinson’s disease, ataxias, ALS, and multiple sclerosis.
Probing molecular, cellular changes underlying pathology
In addition to his leadership role, Dr. Thinakaran oversees a laboratory at the Byrd Alzheimer’s Center where he uses cutting-edge cell biology techniques and mouse models to study the molecular and cellular processes underlying Alzheimer’s disease. His research is supported by more than $6.1 million in grants from the National Institutes of Health (NIH), National Institute on Aging.
With normal brain aging, people experience minor lapses of memory (i.e., forgetting where their keys were left, or the name of someone just met) and some reduced speed in processing information. But disruptions in attention, memory, language, thinking and decision-making that interfere with daily life are signs of dementia.

In addition to overseeing his own laboratory research, Dr. Thinakaran holds Morsani College of Medicine leadership roles as associate dean of neuroscience research and Neuroscience Research Institute associate director of research.
Dr. Thinakaran’s lab pursues findings on relatively new genes identified through genome-wide association studies to gain insights into the mechanisms of late-onset Alzheimer’s disease, which affects people age 65 or older and accounts for the overwhelming majority of cases. Recently, the group has been investigating the role of bridging integrator 1 (BIN1), the second most common genetic risk factor for late-onset Alzheimer’s (exceeded only by APOE). Approximately 40% of people with Alzheimer’s have one of three variations in the BIN1 gene – a glitch in a single DNA building block (nucleotide) that heightens their risk for the disease, Dr. Thinakaran said.
Pursuing a common risk factor for late-onset Alzheimer’s
BIN1, expressed in all the body’s cells, has been shown to play a role in suppressing tumors and in muscle development — but little is known about what the protein does in the brain. Dr. Thinakaran was among the first to embrace the challenge of pursuing how BIN1 contributes to Alzheimer’s disease risk at a time when most researchers focused on amyloid and tau, two proteins considered the primary drivers of Alzheimer’s pathology.
Now, his team and a few others across the country probe what goes wrong in Alzheimer’s patients who carry the BIN1 risk allele. They have already confirmed that BIN1 is present both in the brain’s nerve cells (neurons) and its non-neuronal cells, such as oligodendrocytes and microglia.
A healthy human brain contains tens of billions of neurons that process and transmit chemical messages (neurotransmitters) across a tiny gap between neurons called a synapse. Alzheimer’s disease severely disrupts this synaptic communication, eventually killing cells throughout the brain and leading to a steep decline in memory and other signs of dementia.
“The single biggest correlation with cognitive decline is the loss of these synaptic communication centers between neurons,” Dr. Thinakaran said, adding that individuals most susceptible to developing full-blown Alzheimer’s in later life are those who lose the most synapses.
In a study published March 10 in Cell Reports, Dr. Thinakaran and colleagues demonstrated for the first time that the loss of BIN1 expression impaired spatial learning and memory associated with remembering where things are located. The researchers used an Alzheimer’s disease “knockout” mouse model in which neuronal BIN1 expression was inactivated in the hippocampus, a brain region involved with higher cognitive functions.
Discovering a defect in brain cell communication
A lack of BIN1 leads to a defect in the transmission of neurotransmitters needed to activate the brain cell communication that allows us to think and behave, the researchers found. Further analysis found that BIN1 was primarily located in neurons that send neurotransmitters across the synapse (presynaptic sites) rather than residing on those neurons that receive the neurotransmitter messages (postsynaptic sites). The BIN1 deficiency was also associated with reduced synapse density; a back-up of docked vesicles, the tiny bubble-like carriers that transfer neurotransmitters from presynaptic to postsynaptic neurons; and likely slower release of the neurotransmitters from their vesicles.
“Our findings so far that BIN1 localizes right at the point of (presynaptic) communication and may be precisely regulating neurotransmitter vesicle release brings us much closer to understanding how BIN1 could exert its function as a risk factor (for Alzheimer’s disease),” Dr. Thinakaran said. “We suspect it helps control how efficiently neurons communicate.”

Peering into the brain, one synapse at a time. Electron micrograph depicting selected region of a mouse brain hippocampus, the brain area responsible for learning and memory. A single synapse is marked with the yellow outline. The human brain is estimated to have trillions of these synapses, which transmit information from one neuron to the next.| Image courtesy of Gopal Thinakaran, PhD
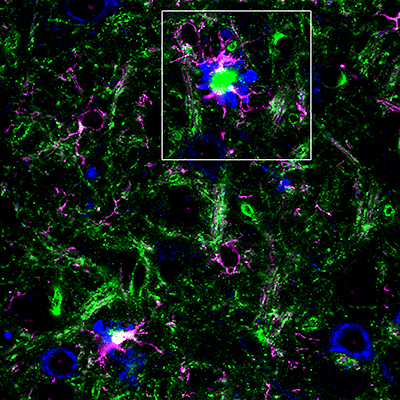
Antibody-stained mouse brain with Alzheimer’s disease β-amyloid deposits. The amyloid precursor proteins within healthy nerve cells and swollen neuronal processes are depicted in blue. The late-onset Alzheimer’s risk factor BIN1 is shown in green, and a marker for brain glial cells responsible for neuroinflammation is shown in magenta.| Image courtesy of Gopal Thinakaran, PhD
Dr. Thinakaran’s team also became interested in investigating whether BIN1 risk variants can interfere with the protective capacity of glia (cells supporting neurons) to mount a full inflammatory response needed to clear toxins from the brain. His USF Health group will work with researchers at Emory University to further investigate why the absence of BIN1 may impair the brain’s removal of abnormal beta-amyloid protein associated with Alzheimer’s disease.
Exploring the type 2 diabetes connection
Collaborating with a coprincipal investigator at the University of Kentucky, Dr. Thinakaran also explores the molecular link between type 2 diabetes and Alzheimer’s disease progression. An Alzheimer’s mouse model created by the Thinakaran lab allows researchers to turn on, or switch off, production of the human hormone amylin in the pancreas.
Amylin is secreted by the pancreas at higher levels, along with insulin, as diabetes begins to develop. Small amounts of this excess amylin migrate from pancreatic cells into the bloodstream and can cross the blood-brain barrier, especially in older brains where the protective barrier becomes leakier. The amylin then mixes with the brain’s beta-amyloid, which eventually builds into the sticky amyloid plaques that are a hallmark of Alzheimer’s pathology. The researchers will test in their preclinical model whether this brain amylin elevates the risk for Alzheimer’s disease, and if reducing amylin in peripheral circulation can help prevent or slow damage to cognition.
Scientists are still trying to figure out why some people remain cognitively resilient throughout life despite having neuropathology that would otherwise cause dementia. On the horizon, Dr. Thinakaran said, integrating large databases of gene expression and individual cell types will help scientists drill deeper into what specific inflammatory, metabolic and neural circuit changes shift a normally aging brain to one in which the abilities to remember, think and reason abnormally accelerate.
At the same time, data on genetics and environment/lifestyle (including diet, physical and mental exercises, sleep patterns and uncontrolled cardiovascular risk factors such as hypertension, diabetes and high cholesterol) are being collected both for patients in various stages of Alzheimer’s disease and for older adults with healthy cognitive function. “Bridging these two sets of data will be extremely valuable in understanding what confers higher risk and delineating what can keep our brains healthy as we age,” Dr. Thinakaran said.
Fascinated by a field with unprecedented challenges
Dr. Thinakaran holds a PhD in molecular biology and genetics from the University of Guelph in Canada. He completed a postdoctoral research fellowship in neuropathology and was an assistant professor of pathology at Johns Hopkins University School of Medicine. Before joining USF Health, he was a professor of neurobiology at the University of Chicago, where he built one of the country’s leading laboratories investigating pathways responsible for Alzheimer’s disease pathology and neuronal dysfunction.
Known as an accomplished scientist and thought leader who does not hesitate to tackle uncharted territory, Dr. Thinakaran studied muscle differentiation as a PhD student. But, he soon realized that muscle research had advanced to a stage where it was unlikely he could make much of an impact. At that time (early 1990s) Alzheimer’s disease research was just gaining momentum in molecular and cellular biology and posing unprecedented challenges, he said.
Once Dr. Thinakaran’s interest in Alzheimer’s was sparked during his postdoctoral training at Johns Hopkins, he seized the opportunity to pursue the emerging area of neuroscience research. “In many ways the brain and its complexity as we age is the final frontier in understanding human behavior. We’re continuing to learn every day the basics of how this organ system works, and what goes wrong when it doesn’t,” he said. “It’s a field that still has great opportunities for the next generation of young minds to make a difference.”
Dr. Thinakaran has authored more than 140 peer-reviewed publications. He is associate editor for the journals Molecular Neurodegeneration and Genes and Diseases and an editorial board member for Neurodegenerative Diseases and for Current Alzheimer Research. He serves on several scientific review/advisory committees for federal, private and public institutions. Dr. Thinakaran has received numerous awards, including the Alzheimer’s Association prestigious Zenith Fellows Award supporting senior scientists pursuing new ideas to advance Alzheimer’s and dementia research.
Some things you may not know about Dr. Thinakaran
- Dr. Thinakaran combines his artistic talents of drawing and painting with his research. Andy Warhol-like microscopic art he created won a competition and was featured as the program cover for a brain research symposium at the University of Chicago. The multicolor montage of images depicts a mouse brain section (hippocampus) stained to visualize β-secretase, an enzyme critical for generating the hallmark Alzheimer’s disease β-amyloid pathology.
- He is married to neurophysiologist Angèle Parent, PhD, associate professor of molecular medicine at the Byrd Alzheimer’s Center. They have three children: Abigaël, a freshman and aspiring neuroscientist at the University of Chicago; Daphné, 14; and Cédric, 12.
- Dr. Thinakaran enjoys cooking authentic South Indian food and other international dishes with his family.
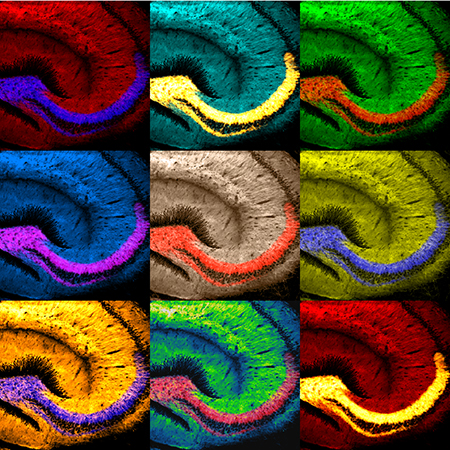
This microscopic brain art created by Dr. Thinakaran was featured on the program cover of a University of Chicago brain research symposium.
-Video by Allison Long, and photos by Freddie Coleman, USF Health Communications and Marketing






